Toning / metallic toning (French: virage, German: Tonung)
Description
In contrast to tinting, toning is not the simple immersion of a film into a dye bath but involves a chemical reaction converting the silver image. In this reaction the neutral silver image in the emulsion of the positive film is replaced by one consisting of colored metal compounds. These were usually iron ferrocyanide (Prussian Blue) for blue, copper ferrocyanide for red/brown, silver sulfide for sepia or rarely uranium ferrocyanide for reddish brown. Toning had been used in still photography before. But since film was projected on the screen it required translucent toning compounds.
In principle, toned images can be identified easily by the transparent bright image parts and perforation areas, while the shadows and the dark parts are colored in a specific hue (see images on this page). In practice, however, the brighter parts can also be affected by the diffusion of dyeing compounds, or the images were tinted and toned in combination.
There were two chemical recipes available for toning, either a one-bath or a two-bath process. In the one-bath process, the silver image is converted to silver ferrocyanide and then combined with the colored ferrocyanide. In the two-bath process, metallic ferricyanide first bleaches the metal silver to silver ferrocyanide. In the second step, the silver ferrocyanide is converted to colored metal ferrocyanide.
See also mordant or dye toning
-
![]()
Hongarije (FRA 1926, Anonymous). Credit: EYE Film Museum. Photographs of the tinted, toned and stencil colored nitrate print by Olivia Kristina Stutz, ERC Advanced Grant FilmColors.
-
![]()
Virages sur films à support teinté Pathé, Film teinté lavande (virage bleu) lavender tinted stock with blue toning, the same image from a different copy of the book, combination of tungsten backlight with daylight toplight. Photograph by Barbara Flueckiger. Source: Didiée, L. (1926): Le Film vierge Pathé. Manuel de développement et de tirage. Paris: Pathé.
-
![]()
Photomicrograph, 25x. Credit: Norbert Wey, Institute of Pathology, University of Zurich.
-
![]()
Photomicrograph, 50x. Credit: Norbert Wey, Institute of Pathology, University of Zurich.
-
![]()
Photomicrograph, 100x. Credit: Norbert Wey, Institute of Pathology, University of Zurich.
-
![]()
Toning samples from the Tinting and Toning Workshop by Ulrich Ruedel, Seminar “Materiality of Film” by Barbara Flueckiger and Bregt Lameris, Department of Film Studies, University of Zurich, in collaboration with Lichtspiel / Kinemathek Bern, David Landolf and Brigitte Paulowitz.
-
![]()
Toning samples from the Tinting and Toning Workshop by Ulrich Ruedel, Seminar “Materiality of Film” by Barbara Flueckiger and Bregt Lameris, Department of Film Studies, University of Zurich, in collaboration with Lichtspiel / Kinemathek Bern, David Landolf and Brigitte Paulowitz.
Galleries Open all Galleries ▼
Original Technical Papers and Primary Sources
(1925*): Agfa Kine-Handbuch. Berlin: Actien-Gesellschaft für Anilin-Fabrikation, Teil I on pp. 79–80 and Teil III on pp. 7–15. (in German) [* year estimated] View Quote
Anonymous (1911): Toning and Tinting as an Adjunct to the Picture. In: Moving Picture World, 8, 18.3.1911, p. 574. View Quote
Anonymous (1937): Toning Not Tinting, Used on Good Earth and Winkie Release Prints. In: International Projectionist, 12,9, pp. 22–23. View Link
Bedding, Thomas (1909): Practical Moving-Picture Making. Tinting and Toning. In: The Bioscope, 157, Oct., pp. 23–25. View Quote
Conrad-Alberti, Victor (1933): Die technischen und künstlerischen Voraussetzungen für die Herstellung farbiger Kulturfilme. In: Film-Kurier, 112, 13.5.1933.) (in German) View Quote
Didiée, L. (1926): Le Film vierge Pathé. Manuel de développement et de tirage. Paris: Pathé, pp. 129-135. (in French) View Quote
Duncan, F. Martin (1909): Artistic Toning Effects. In: The Bioscope, 130, Apr., p. 17. View Quote
Duncan, F. Martin (1909): Artistic Toning Effects. In: The Bioscope, 131, Apr., p. 15. View Quote
Eastman Kodak Company (1916): Tinting and Toning of Eastman Positive Motion Picture Film. Rochester, NY. [following editions in 1918, 1922, 1924, and 1927] View Quote
Eastman Kodak Company (1927): Tinting and Toning of Eastman Positive Motion Picture Film. Rochester, NY: Eastman Kodak, on p. 6 View Quote, on pp. 13–14 View Quote, on pp. 17–18 View Quote, on p. 18 View Quote, on p. 23 View Quote, on pp. 23–24 View Quote, on p. 24 View Quote, on p. 25 View Quote, on pp. 25–26 View Quote, on p. 26 View Quote, on pp. 26–27 View Quote, on pp. 27–28 View Quote, on pp. 28–30 View Quote, on pp. 30–32 View Quote, on p. 32 View Quote, on pp. 32–34 View Quote, on p. 35 View Quote, on pp. 35–36 View Quote, on p. 36 View Quote, on pp. 36–39. View Quote
Fischer, Rudolf (1913): Toning Bromide Prints. In: The British Journal of Photography, 60, p. 712. View Quote
Nickolaus, J. M. (1937): Toning Positive Film by Machine Methods. In: Journal of the Society of Motion Picture Engineers, 29,1,Jul., pp. 65–67. View Link
Secondary Sources
Abel, Richard (1996): Pathé’s ‘Heavenly Billboards’. In: Monica Dall’Asta, Guglielmo Pescatore and Leonardo Quaresima (eds.): Il colore nel cinema muto. Bologna: Clueb, pp. 56–76, on p. 60. View Quote
Abel, Richard (1996): Pathé’s ‘Heavenly Billboards’. In: Monica Dall’Asta, Guglielmo Pescatore and Leonardo Quaresima (eds.): Il colore nel cinema muto. Bologna: Clueb, pp. 56–76, on p. 61. View Quote
Alt, Dirk (2011): “Der Farbfilm marschiert!” Frühe Farbfilmverfahren und NS-Propaganda 1933-1945. München: Belleville, on pp. 36–37. (in German) View Quote
Basten, Fred E. (1980): Glorious Technicolor. The Movies’ Magic Rainbow. South Brunswick: Barnes, on pp. 14–15. View Quote
Brown, Simon (2012): Technical Appendix. In: Sarah Street: Colour Films in Britain. The Negotiation of Innovation 1900-55. Basingstoke, Hampshire: Palgrave Macmillan, pp. 259-287, on pp. 286-287. View Quote
Borde, Raymond (1988): Die Filmarchive und der Farbfilm. Eine Einführung. In: Gert Koshofer: Color. Die Farben des Films. Berlin: Wissenschaftsverl. Volker Spiess, pp. 7–10, on p. 8. (in German) View Quote
Cherchi Usai, Paolo (1996): The Color of Nitrate. Some Factual Observations on Tinting and Toning Manuals for Silent Films. In: Abel, Richard (Hrsg): Silent Film. New Brunswick : Rutgers University Press, pp. 21-30, on pp. 26-27. View Quote
Cherchi Usai, Paolo (2000): Silent Cinema. London: BFI, pp. 24-27. View Quote
Cleveland, David; Pritchard, Brian (2015): How Films were Made and Shown. Some Aspects of the Technical Side of Motion Picture Film 1895-2015. Manningtree, Essex: David Cleveland, on pp. 195–196. View Quote
De Kuyper, Eric (1992): La beauté du diable. In: Michel Ciment (ed.): Ciné mémoire. Colloque international d’information (7-9 octobre 1991). Paris: Femis, pp. 29–31. (in French) View Quote
Degenhardt, Inge (1996): On the Absence and Presence of Colour in Film. In: Monica Dall’Asta, Guglielmo Pescatore and Leonardo Quaresima (eds.): Il colore nel cinema muto. Bologna: Clueb, pp. 88–102. View Quote
Eggert, John; Heymer, Gerd (1937): Der Stand der Farbenphotographie. In: Veröffentlichungen des wissenschaftlichen Zentral-Laboratoriums der photographischen Abteilung Agfa, pp. 7–28, on p. 21. (in German) View Quote
Everett, Wendy (2007): Mapping Colour. An Introduction to the Theories and Practices of Colour. In: Wendy Everett (ed.): Questions of Colour in Cinema. From Paintbrush to Pixel. Oxford: Peter Lang, pp. 7–38, on p. 18 View Quote and on pp. 18–19. View Quote
Famulener, K. (1939): Some Studies on the Use of Color Coupling Developers for Toning Processes. In: Journal of the Society of Motion Picture Engineers, 4, pp. 412–418, on pp. 412–414 and on p. 417. View Quote
Fossati, Giovanna (1998): When Cinema Was Coloured. In: Luciano Berriatúa et al.: Tutti i colori del mondo. Il colore nei mass media tra 1900 e 1930. = All the colours of the world. Reggio Emilia: Edizioni Diabasis, pp. 121-132, on pp. 125-126 View Quote, pp. 127-128 View Quote and p. 130 View Quote.
Genaitay, Sonia; Dixon, Bryony (2010): Early Colour Film Restoration at the BFI National Archive. In: Journal of British Cinema and Television, 7,1, pp. 131-146.
Goergen, Jeanpaul (2010): Bunte Bilder aus dem Farbenbottich. Tonung und Virage in dokumentarischen Filmen der 1910er und 1920er Jahre. In: Filmblatt, pp. 3–52, on pp. 3–4 View Quote, on pp. 5–7 View Quote, on p. 10 View Quote, on pp. 15–20 View Quote, on p. 43 View Quote and on p. 44–45. (in German) View Quote
Layton, James; Pierce, David (2015): The Dawn of Technicolor. Rochester: George Eastman House, on pp. 111–112. View Quote
Mazzanti, Nicola (2009): Colours, Audiences, and (Dis)Continuity in the ‘Cinema of the Second Period’. In: Film History, 21,1, 2009, pp. 67-93. Link to download.
Montesanti, Fausto (1954): Lineamenti di una storia del film a colori. In: Giuseppe Sala (ed.): Bianco e Nero. Il colore nel cinema. Rassegna mensile di studi cinematografici, XV,2-4, pp. 11–51, on p. 18 View Quote and on pp. 19–22. (in Italian) View Quote
Oliveira, João S. de (2002): Black-and-White in Colour. In: Roger Smither (ed.), This Film is Dangerous. A Celebration of Nitrate Film. Brussels: FIAF, pp. 117-122, on pp. 118-119. View Quote
Pierotti, Federico (2012): La seduzione dello spettro. Storia e cultura del colore nel cinema. Genova: Le Mani-Microart, on p. 20 View Quote and on pp. 52–53. (in Italian) View Quote
Pinel, Vincent (1992): La forêt des techniques. In: Michel Ciment (ed.): Ciné mémoire. Colloque international d’information (7-9 octobre 1991). Paris: Femis, pp. 17-24, on pp. 18-19. (in French) View Quote
Read, Paul (1998): Tinting and Toning Techniques and their Adaption for the Restoration of Archive Film. In: Luciano Berriatúa et al. (eds.): Tutti i colori del mondo. Il colore nei mass media tra 1900 e 1930. = All the colours of the world. Reggio Emilia: Diabasis, pp. 157–167, on pp. 157–158. View Quote
Read, Paul (2009): ‘Unnatural Colours’. An Introduction to Colouring Techniques in Silent Era Movies. In: Film History, 21.1, pp. 9-46, on pp. 14-15 View Quote, see list of metallic tones on pp. 37-39 View Quote and combined tones on pp. 44-45. View Quote
Ruivo, Céline (2013): Le Livre de fabrication de la compagnie générale des phonographes cinématographes et appareils de précision. À propos d’une source pour l’histoire des recherches sur la couleur chez Pathé Frères entre 1906 et 1908. In: 1895. Revue d’Histoire du Cinéma, 71, pp. 47–60, on pp. 51–53 View Quote, on pp. 54–56 View Quote, on p. 56 View Quote and on pp. 56–58. (in French) View Quote
Ryan, Roderick T. (1977): A History of Motion Picture Color Technology. London: Focal Press, pp. 19-20. View Quote
Stokes, Melvyn (2009): Colour in American Cinema. From The Great Train Robbery to Bonnie and Clyde. In: Raphaëlle Costa de Beauregard (ed.): Cinéma et couleur. Paris: M. Houdiard, pp. 184–192, on p. 185. View Quote
Street, Sarah; Yumibe, Joshua (2019): Chromatic Modernity. Color, Cinema, and Media of the 1920s. New York: Columbia University Press, on pp. 34–35. View Quote
Yumibe, Joshua (2012): Moving Colors. Early Film, Mass Culture, Modernism. New Brunswick et al.: Rutgers University Press, pp. 4-6 View Quote and pp. 100-105. View Quote
Edge Codes and Identification
Brown, Harold (1990): Physical Characteristics of Early Films as Aids to Identification. Brussels: FIAF.
Agfa: Edge marks. View Quote on Page: Edge Codes and Identification
View Image in Gallery: Roald Amundsen’s North Pole Expedition (Norway 1924)
View Image in Gallery: Roald Amundsen’s North Pole Expedition (Norway 1924)
View Image in Gallery: Roald Amundsen’s North Pole Expedition (Norway 1924)
View Image in Gallery: Roald Amundsen’s North Pole Expedition (Norway 1924)
View Image in Gallery: Roald Amundsen’s North Pole Expedition (Norway 1924)
View Image in Gallery: Der Alpenweg. Sallanches, Gombloux, Megeve (CH [1920])
View Image in Gallery: Die Hochbahnkatastrophe (GER 1921)
Cines: Edge marks. View Quote on Page: Edge Codes and Identification
View Image in Gallery: Massafra (ITA 1911)
View Image in Gallery: Massafra (ITA 1911)
View Image in Gallery: Massafra (ITA 1911)
View Image in Gallery: Massafra (ITA 1911)
Cines: Frame characteristics / features of margins. View Quote on Page: Edge Codes and Identification
Cines: Title styles. View Quote on Page: Edge Codes and Identification
Eclair: Edge marks. View Quote on Page: Edge Codes and Identification
View Image in Gallery: Comment le soleil se couche (FRA 1913)
View Image in Gallery: Comment le soleil se couche (FRA 1913)
View Image in Gallery: Comment le soleil se couche (FRA 1913)
Eclair: Title styles. View Quote on Page: Edge Codes and Identification
View Image in Gallery: Comment le soleil se couche (FRA 1913)
View Image in Gallery: Comment le soleil se couche (FRA 1913)
View Image in Gallery: Comment le soleil se couche (FRA 1913)
View Image in Gallery: Comment le soleil se couche (FRA 1913)
Gaumont: Edge marks. View Quote on Page: Edge Codes and Identification
View Image in Gallery: [Gaumont Fragments]
View Image in Gallery: [Gaumont Fragments]
View Image in Gallery: [Gaumont Fragments]
View Image in Gallery: [Gaumont Fragments]
View Image in Gallery: [Gaumont Fragments]
View Image in Gallery: [Gaumont Fragments]
View Image in Gallery: [Gaumont Fragments]
Gevaert: Edge marks. View Quote on Page: Edge Codes and Identification
View Image in Gallery: Voyage autour d’une étoile (FRA 1906, Gaston Velle)
Kodak: Edge marks. View Quote on Page: Edge Codes and Identification
View Image in Gallery: Das Cabinet des Dr. Caligari (1919)
View Image in Gallery: Das Cabinet des Dr. Caligari (1919)
View Image in Gallery: Grass. A Nation’s Battle for Life (USA 1925)
View Image in Gallery: Grass. A Nation’s Battle for Life (USA 1925)
View Image in Gallery: Grass. A Nation’s Battle for Life (USA 1925)
View Image in Gallery: Malombra (ITA 1917)
View Image in Gallery: Malombra (ITA 1917)
View Image in Gallery: Malombra (ITA 1917)
View Image in Gallery: Malombra (ITA 1917)
View Image in Gallery: Der Alpenweg. Sallanches, Gombloux, Megeve (CH [1920])
View Image in Gallery: Toning Samples (Kodak Film Samples Collection)
View Image in Gallery: Toning Samples (Kodak Film Samples Collection)
Pathé: Edge marks (producer). View Quote on Page: Edge Codes and Identification
View Images in Gallery: An Atlantic Voyage (GER / GBR / FRA)
View Images in Gallery: Les petits vagabonds (FRA 1905, Lucien Nonguet)
Pathé: Edge marks (stock manufacturer). View Quote on Page: Edge Codes and Identification
View Image in Gallery: [Tänze] (? 1920s)
View Image in Gallery: [Tänze] (? 1920s)
View Image in Gallery: [Tänze] (? 1920s)
View Image in Gallery: [Tänze] (? 1920s)
View Image in Gallery: [Tänze] (? 1920s)
View Image in Gallery: [Tänze] (? 1920s)
View Image in Gallery: [Tänze] (? 1920s)
Measurements
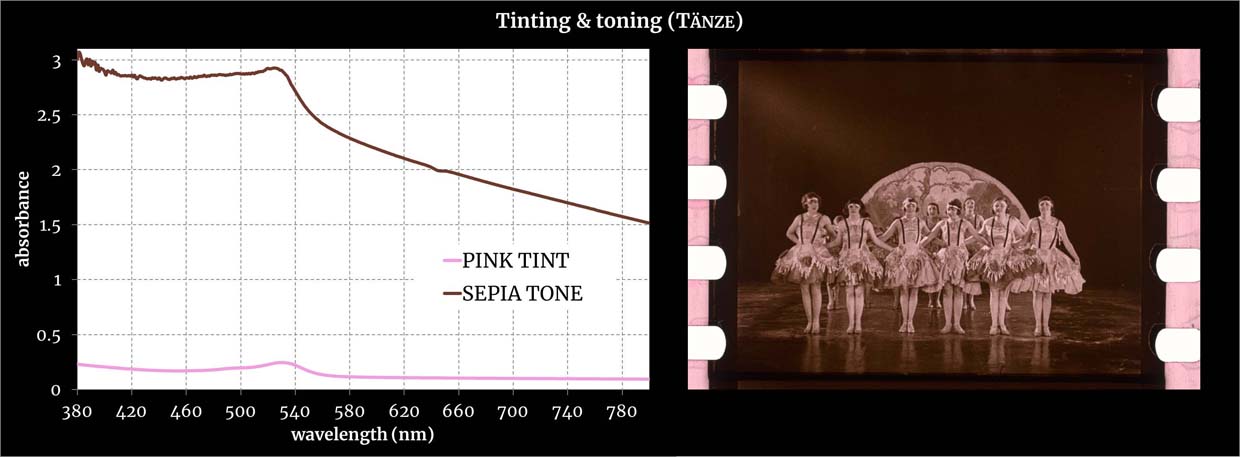
Absorbances measured with a double-beam spectrophotometer from film areas with pure dyes. Credit: Giorgio Trumpy, ERC Advanced Grant FilmColors.
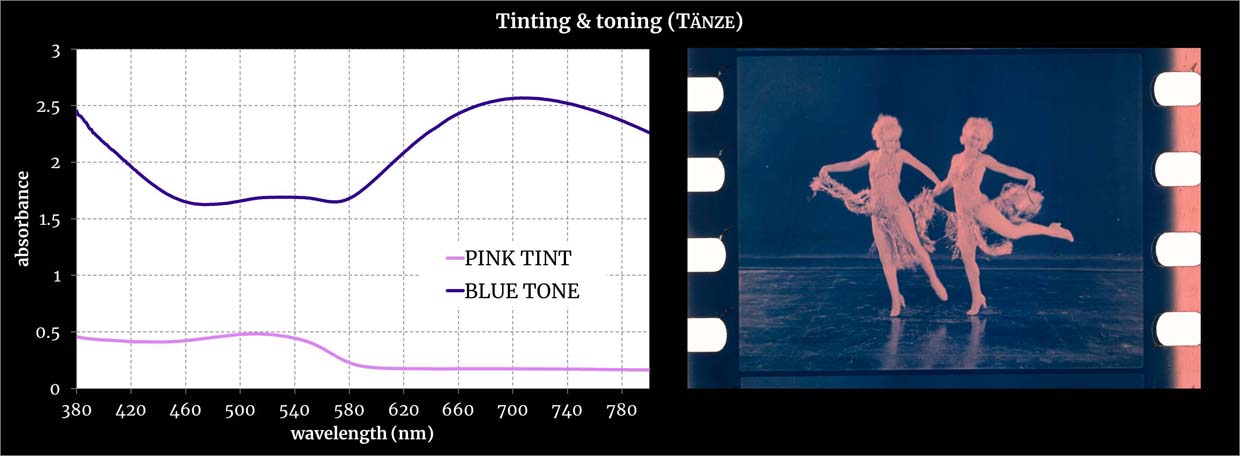
Absorbances measured with a double-beam spectrophotometer from film areas with pure dyes. Credit: Giorgio Trumpy, ERC Advanced Grant FilmColors.
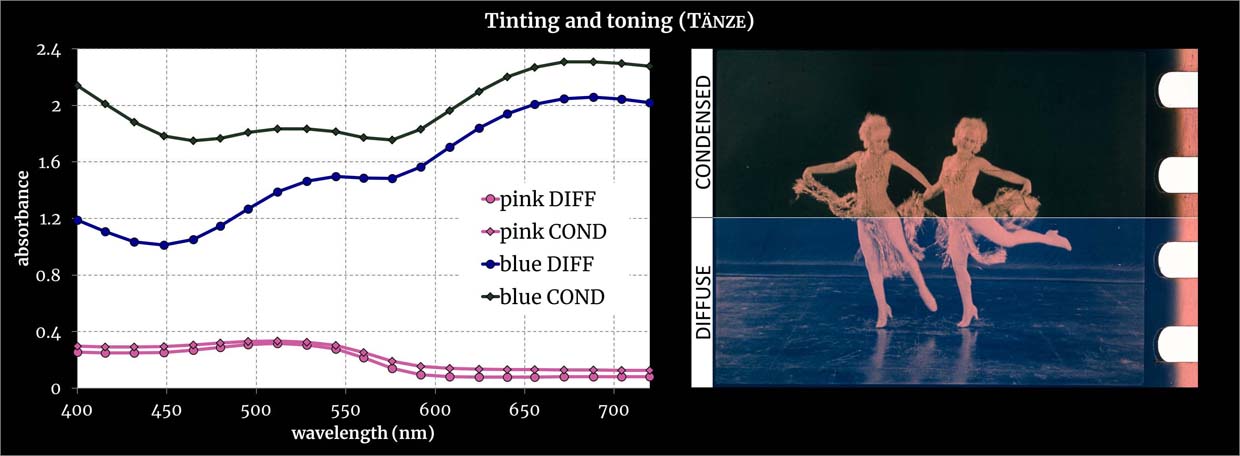
Multispectral acquisition in condensed and diffuse illumination. For both light configurations, absorbances of deep blue toning and pink tinting (left) and colorimetric images (right). Credit: Giorgio Trumpy, ERC Advanced Grant FilmColors.
See the specific sample from which the spectra were measured.
Films
Jeanne d’Arc (FRA 1900, Georges Méliès) (sequences)22
La vie et la passion de Jésus Christ (FRA 1903, Ferdinand Zecca/Lucien Nonguet)22
La poule aux oeufs d’or (FRA 1905, Gaston Velle)5
La Vie au Japon, les rapides de la rivière Ozu (FRA 1906, André Legrand) (sequences)3
Die Verräterin (GER 1911, Urban Gad) (sequences)12
Inferno (ITA 1911, Francesco Bertolini, Giuseppe De Liguoro, Adolfo Padovan) (sequences)9
Intolerance (USA 1916, D. W. Griffith) (sequences)19
Joan the Woman (USA 1916, Cecil B. DeMille) (sequences)1
Rapsodia satanica (ITA 1917, Nino Oxilia) (sequences)2
Das Cabinet des Dr. Caligari (GER 1919, Robert Wiene) (sequences)8, 18, 19
L’Atlantide/Queen of Atlantis (FRA/BEL 1921, Jacques Feyder) (sequences)12
Nosferatu. Eine Symphonie des Grauens (GER 1922, Friedrich Wilhelm Murnau) (sequences)8, 13, 19
The Ten Commandments (USA 1923, Cecil B. DeMille) (sequences)4
The Great White Silence (GBR 1924, Herbert G. Ponting) (sequences)20
The Phantom of the Opera (sequences) (U, USA 1925, Rupert Julian)6, 8, 16, 21
The Son of the Sheik (USA 1926, George Fitzmaurice) (sequences)14
Metropolis (GER 1927, Fritz Lang) (sequences)15
Napoléon (FRA 1927, Abel Gance) (sequences)8, 17
The King of Kings (sequences) (Pathe, USA 1927, Cecil B. DeMille)7
The Lodger – A Story of the London Fog (GBR 1927, Alfred Hitchcock) (sequences)11, 17, 20
Wee Willie Winkie (USA 1937, John Ford) (sequences)10
Le Bagne des gosses (FRA 1907, Charles Decroix)23
Nuit de Noël (FRA 1908)23
La vengeance du forgeron (FRA 1907, Lucien Nonguet)23
Les Forbans (FRA 1907)23
1 Pierotti, Federico (2012): La seduzione dello spettro. Storia e cultura del colore nel cinema. Genova: Le Mani-Microart, on pp. 96–97. (in Italian)
2 Pierotti, Federico (2012): La seduzione dello spettro. Storia e cultura del colore nel cinema. Genova: Le Mani-Microart, on pp. 76–79. (in Italian)
3 Ruivo, Céline (2013): Le Livre de fabrication de la compagnie générale des phonographes cinématographes et appareils de précision. À propos d’une source pour l’histoire des recherches sur la couleur chez Pathé Frères entre 1906 et 1908. In: 1895. Revue d’Histoire du Cinema, 71, 2013, pp. 47–60, on p. 56. (in French) View Quote
4 Layton, James; Pierce, David (2015): The Dawn of Technicolor. Rochester: George Eastman House, on pp. 99–102.
5 Pierotti, Federico (2012): La seduzione dello spettro. Storia e cultura del colore nel cinema. Genova: Le Mani-Microart, on pp. 43–47.) (in Italian)
6 Kitsopanidou, Kira (2009): “Glorious Technicolor”. La stratégie d’innovation de la couleur de Technicolor dans l’industrie cinématographique Américaine. In: Raphaëlle Costa de Beauregard (ed.): Cinéma et couleur. Paris: M. Houdiard, pp. 193–206, on p. 194. (in French)
7 Behlmer, Rudy (1964): Technicolor. In: Films in Review, 15,6, pp. 333–351, on pp. 337–342.
8 Traber, Bodo (1995): Dramaturgische Funktionen monochromer Farbgebung im Stummfilm. In: Karl-Dietmar Möller-Nass Möller-Nass, Hasko Schneider and Hans J. Wulff (eds.): 1. Film- und Fernsehwissenschaftliches Kolloquium. Münster: MAkS, pp. 30–36, on pp. 30–35. (in German)
9 Pierotti, Federico (2016): Un’archeologia del colore nel cinema italiano. Dal Technicolor ad Antonioni. Pisa: Edizioni ETS, on pp. 64–69. (in Italian)
10 Heckman, Heather (2013): The Cameraman and the Glamour-Puss. Technicolor Cinematography and Design in John Ford’s Drums Along the Mohawk. In: Brown, Simon; Street, Sarah; Watkins, Liz: Color and the Moving Image. History, Theory, Aesthetics, Archive. New York, London: Routledge, pp. 160–169.
11 Read, Paul (1998): Tinting and Toning Techniques and their Adaption for the Restoration of Archive Film. In: Luciano Berriatúa et al. (eds.): Tutti i colori del mondo. Il colore nei mass media tra 1900 e 1930. = All the colours of the world. Reggio Emilia: Diabasis, pp. 157‒167, on pp. 160–161. View Quote
12 De Kuyper, Eric (1992): La beauté du diable. In: Michel Ciment (ed.): Ciné mémoire. Colloque international d’information (7-9 octobre 1991). Paris: Femis, pp. 29–31. (in French) View Quote
13 Watkins, Liz (2013): Interview. Giovanna Fossati. In: Simon Brown, Sarah Street and Liz Watkins (eds.): British Colour Cinema. Practices and Theories. Hampshire: Palgrave Macmillan, pp. 228–235, on pp. 229–235. View Quote
14 Traber, Bodo (1995): Dramaturgische Funktionen monochromer Farbgebung im Stummfilm. In: Karl-Dietmar Möller-Nass Möller-Nass, Hasko Schneider and Hans J. Wulff (eds.): 1. Film- und Fernsehwissenschaftliches Kolloquium. Münster: MAkS, pp. 30–36, on pp. 32–35. (in German) View Quote
15 Watkins, Liz (2013): Interview. Paolo Cherchi Usai. In: Simon Brown, Sarah Street and Liz Watkins (eds.): British Colour Cinema. Practices and Theories. Hampshire: Palgrave Macmillan, pp. 208–218, on pp. 215–218. View Quote
16 Traber, Bodo (1995): Dramaturgische Funktionen monochromer Farbgebung im Stummfilm. In: Karl-Dietmar Möller-Nass Möller-Nass, Hasko Schneider and Hans J. Wulff (eds.): 1. Film- und Fernsehwissenschaftliches Kolloquium. Münster: MAkS, pp. 30–36, on pp. 31–33. (in German) View Quote
17 Watkins, Liz (2013): Interview. João S. De Oliveira, Hon. FBKS. In: Simon Brown, Sarah Street and Liz Watkins (eds.): British Colour Cinema. Practices and Theories. Hampshire: Palgrave Macmillan, pp. 171–185, on pp. 175–185. View Quote
18 Degenhardt, Inge (1996): On the Absence and Presence of Colour in Film. In: Monica Dall’Asta, Guglielmo Pescatore and Leonardo Quaresima (eds.): Il colore nel cinema muto. Bologna: Clueb, pp. 88–102. View Quote
19 Gunning, Tom (2013): Where Do Colors Go at Night? In: Simon Brown, Sarah Street and Liz Watkins (eds.): Color and the Moving Image. History, Theory, Aesthetics, Archive. New York, London: Routledge, pp. 81–92, on pp. 82–86. View Quote
20 Watkins, Liz (2013): Herbert G. Ponting’s Materials and Texts. In: Simon Brown, Sarah Street and Liz Watkins (eds.): Color and the Moving Image. History, Theory, Aesthetics, Archive. New York, London: Routledge, pp. 230–242, on pp. 230–240. View Quote
21 Higgins, Scott (2007): Harnessing the Technicolor Rainbow. Color Design in the 1930s. Austin: University of Texas Press, on pp. 3–4.
22 Montesanti, Fausto (1954): Lineamenti di una storia del film a colori. In: Giuseppe Sala (ed.): Bianco e Nero. Il colore nel cinema. Rassegna mensile di studi cinematografici, XV,2-4, pp. 11–51, on pp. 14–17. (in Italian)
23 Abel, Richard (1996): Pathé’s ‘Heavenly Billboards’. In: Monica Dall’Asta, Guglielmo Pescatore and Leonardo Quaresima (eds.): Il colore nel cinema muto. Bologna: Clueb, pp. 56–76, on p. 60. View Quote
Downloads
Eastman Kodak Company (1918): Tinting and Toning of Eastman Positive Motion Picture Film. Rochester, NY.
Download PDFFischer, Rudolf (1913): Toning Bromide Prints – No. 5,602, 1913. In: The British Journal of Photography, 60, p. 712.
Download PDFCrabtree, J.I.; Marsh, W. (1931): Double Toning of Motion Picture Films. In: Journal of the Society of Motion Picture Engineers, Jan. 1931, pp. 57–60.
Download PDFAnonymous (1920): Die kolorierten Filme. Zum Farbenproblem im Film. In: Film-Kurier, 30.9.1920. (in German)
Download PDF
Patents
Links
Anonymous (1937): Toning Not Tinting, Used on Good Earth and Winkie Release Prints. In: International Projectionist, 12,9, pp. 22–23. View Link
The 1922 edition of the Eastman Kodak Manual
Tinting and Toning of Eastman Positive Motion Picture Film.
The 1927 edition of the Eastman Kodak Manual
Tinting and Toning of Eastman Positive Motion Picture Film.
Nickolaus, J. M. (1937): Toning Positive Film by Machine Methods. In: Journal of the Society of Motion Picture Engineers, 29,1,July, pp. 65–67. View Link
A great resource is Brian Pritchard’s website. View Link
A comprehensive listing of metallic tones is available on pp. 37-39 in
Read, Paul (2009): ‘Unnatural Colours’. An Introduction to Colouring Techniques in Silent Era Movies. In: Film History, 21.1, pp. 9-46. View Link
Restoration
Aubert, Michelle (1992): Pour une déontologie de la restauration des films en couleur. In: Michel Ciment (ed.): Ciné mémoire. Colloque international d’information (7-9 octobre 1991). Paris: Femis, pp. 25–28, on pp. 26–27. (in French) View Quote
Berriatúa, Luciano (1998): Regarding a Catalogue of the Tints Used on the Silent Screen. In: Luciano Berriatúa et al.: Tutti i colori del mondo. Il colore nei mass media tra 1900 e 1930. = All the colours of the world. Reggio Emilia: Edizioni Diabasis, pp. 135-139. View Quote
Boarini, Vittorio (1992): La couleur dans le cinéma muet. Des techniques anciennes pour une restauration moderne. In: Michel Ciment (ed.): Ciné mémoire. Colloque international d’information (7-9 octobre 1991). Paris: Femis, pp. 33–36, on pp. 34–36. (in French) View Quote
Desmet, Noël; Read, Paul (1998): The Desmetcolor Method for Restoring Tinted and Toned Films. In: Luciano Berriatúa, Manlio Brusatin, Noël Desmet, Enrico Fornaroli, Giovanna Fossati, Bertrand Lavedrine, Bob Mabberley, Nicola Mazzanti, Ruggero Pierantoni, Paul Read and Sonja Snoek: Tutti i colori del mondo. Il colore nei mass media tra 1900 e 1930. = All the colours of the world. Reggio Emilia: Edizione Diabasis, pp. 147–150. View Quote
Farinelli, Nicola; Mazzanti, Gian Luca (2001): Il restauro. Metodo e tecnica. In: Gian Piero Brunetta (ed.): Storia del cinema mondiale. Vol. 5. Teorie, strumenti, memorie. Bologna: Einaudi, pp. 1119–1174, on p. 1147 View Quote and on p. 1169. (in Italian) View Quote
Gunning, Tom (2013): Where Do Colors Go at Night? In: Simon Brown, Sarah Street and Liz Watkins (eds.): Color and the Moving Image. History, Theory, Aesthetics, Archive. New York, London: Routledge, pp. 81–92, on pp. 82–86. View Quote
Hertogs, Daan; De Klerk, Nico (1996): Disorderly Order. Colours in Silent Film. The 1995 Amsterdam Workshop. Amsterdam: Stichding Nederlands Filmmuseum, on pp. 6-7 View Quote, on pp. 12-14 View Quote, on pp. 18-19 View Quote, on pp. 23-25 View Quote, on pp. 71-76 View Quote and on pp. 87-88. View Quote
Mabberley, Bob; Read, Paul; Snoek, Sonja (1998): Recording and Reproducing the Original Tints and Tones of Quo Vadis. A Technical Case Study. In: Luciano Berriatúa et al.: Tutti i colori del mondo. Il colore nei mass media tra 1900 e 1930. = All the colours of the world. Reggio Emilia: Edizioni Diabasis, pp. 151-155. View Quote
Mazzanti, Nicola (1998): The Colours of the Film d’Arte Italiana. In: Luciano Berriatúa et al.: Tutti i colori del mondo. Il colore nei mass media tra 1900 e 1930. = All the colours of the world. Reggio Emilia: Edizioni Diabasis, pp. 141-146. View Quote
Read, Paul (1998): Tinting and Toning Techniques and their Adaption for the Restoration of Archive Film. In: Luciano Berriatúa et al. (eds.): Tutti i colori del mondo. Il colore nei mass media tra 1900 e 1930. = All the colours of the world. Reggio Emilia: Diabasis, pp. 157‒167, on pp. 160‒161 View Quote, on pp. 161–162 View Quote, on p. 163 View Quote, pp. 163–164 View Quote and on pp. 164–166. View Quote
Read, Paul; Meyer, Mark-Paul (2000): Films Coloured by Toning. In: Restoration of Motion Picture Film. Oxford, pp. 185-188. View Quote
Street, Sarah (2012): Colour Films in Britain. The Negotiation of Innovation 1900-55. Basingstoke, Hampshire: Palgrave Macmillan, on p. 20. View Quote
Traber, Bodo (1995): Dramaturgische Funktionen monochromer Farbgebung im Stummfilm. In: Karl-Dietmar Möller-Nass Möller-Nass, Hasko Schneider and Hans J. Wulff (eds.): 1. Film- und Fernsehwissenschaftliches Kolloquium. Münster: MAkS, pp. 30–36, on pp. 33–34. (in German) View Quote
Watkins, Liz (2013): Herbert G. Ponting’s Materials and Texts. In: Simon Brown, Sarah Street and Liz Watkins (eds.): Color and the Moving Image. History, Theory, Aesthetics, Archive. New York, London: Routledge, pp. 230–242, on pp. 230–240. View Quote
Watkins, Liz (2013): Interview. Giovanna Fossati. In: Simon Brown, Sarah Street and Liz Watkins (eds.): British Colour Cinema. Practices and Theories. Hampshire: Palgrave Macmillan, pp. 228–235, on pp. 229–235. View Quote
Watkins, Liz (2013): Interview. João S. De Oliveira, Hon. FBKS. In: Simon Brown, Sarah Street and Liz Watkins (eds.): British Colour Cinema. Practices and Theories. Hampshire: Palgrave Macmillan, pp. 171–185, on pp. 175–185. View Quote
Watkins, Liz (2013): Interview. Paolo Cherchi Usai. In: Simon Brown, Sarah Street and Liz Watkins (eds.): British Colour Cinema. Practices and Theories. Hampshire: Palgrave Macmillan, pp. 208–218, on pp. 215–218. View Quote
Watkins, Liz (2013): Interview. Ulrich Rüdel and Daniela Currò. In: Simon Brown, Sarah Street and Liz Watkins (eds.): British Colour Cinema. Practices and Theories. Hampshire: Palgrave Macmillan, pp. 219–227, on pp. 221–226. View Quote
The Lodger. A Story of the London Fog (GBR 1926, Alfred Hitchcock):
Heller, Franziska (2015): Alfred Hitchcock. Einführung in seine Filme und Filmästhetik. München: Wilhelm Fink, on pp. 47–48. (in German) View Quote
Contemporary Reception
Cocanari, Silvio (1912): I viraggi. In: La Vita Cinematografica, III,3, 15 febbraio 1912, pp. 2–5 published in Grifo, Marco (2006): Dal film colorato al cinema a colori. In: Michele Canosa, Giulia Carluccio and Federica Villa (eds.): Cinema muto italiano. Tecnica e tecnologia. Volume primo. Roma: Carocci, pp. 188–195. (in Italian) View Quote
Coustet, Ernest (1921): Le cinéma. Paris: Librairie Hachette, on p. 6 View Quote and on pp. 160-162. (in French) View Quote
Fossati, Giovanna (1998): When Cinema Was Coloured. In: Luciano Berriatúa et al.: Tutti i colori del mondo. Il colore nei mass media tra 1900 e 1930. = All the colours of the world. Reggio Emilia: Edizioni Diabasis, pp. 121-132, on pp. 126-131. View Quote
Rühl, Richard (1923): Virage. In: Film-Kurier, 12.2.1923. (in German) View Quote
The Affairs of Anatol (USA 1921, Cecil B. DeMille):
Smith, Agnes (1921): The Screen in Review. The Affairs of Anatol. In: Picture-Play Magazine, XV,1, Sep., p. 58. View Quote
Full text on the Media History Digital Library website. View Link
Whitely Fletcher, Adele (1921): Across the Silversheet. New Screen Plays In Review. In: Motion Picture Magazine, XXII,8, Sep., pp. 68–69, on p. 69. View Quote
Full text on the Media History Digital Library website. View Link
The King of Kings (USA 1927, Cecil B. DeMille; H. B. Warner; Dorothy Cumming; Ernest Torrence; Joseph Schildkraut):
Anonymous (1926): The King of Kings. In: Variety, Dec., n. p. View Quote
Full text on the Variety website. View Link
The Phantom of the Opera (USA 1925, Rupert Julian):
Barr, Al (1925): A Duplex Report on The Phantom. In: Exhibitors Herald, XXII,12, Sep., p. 52. View Quote
Benson, Sally (1925): The Screen in Review. Expensive and Everything. In: Picture-Play Magazine, XXIII,4, Dec., p. 59. View Quote
Hall, Mordaunt (1925): The Screen. A Fantastic Melodrama. In: The New York Times, Sep., p. 15. View Quote
Full text on The New York Times website. View Link
The Ten Commandments (USA 1923, Cecil B. DeMille):
Anonymous (1923): The Screen. In: New York Times, 22 December 1923, p. 8. View Quote
Anonymous (1924): The Ten Commendations of The Ten Commandments. In: Variety, LXXIII,7, Jan., p. 69. View Quote
Anonymous (1924): The Shadow Stage. A Review of the New Pictures. The Ten Commandments. In: Photoplay, XXV,3, Feb., p. 62. View Quote
Selected Analyses
Cabiria (ITA 1914, Giovanni Pastrone):
Pierotti, Federico (2012): La seduzione dello spettro. Storia e cultura del colore nel cinema. Genova: Le Mani-Microart, on p. 71.) (in Italian) View Quote
Cyrano de Bergerac (ITA, FRA 1923, Augusto Genina):
Street, Sarah; Yumibe, Joshua (2019): Chromatic Modernity. Color, Cinema, and Media of the 1920s. New York: Columbia University Press, on pp. 216–217. View Quote
Das Cabinet des Dr. Caligari (GER 1919, Robert Wiene):
De Fries, H. (1920): Raumgestaltung im Film. In: Wasmuths Monatshefte fur Baukunst, 5, 1921, pp. 63–82, on pp. 68–75. (in German) View Quote
Degenhardt, Inge (1996): On the absence and presence of colour in film. In: Monica Dall’Asta, Guglielmo Pescatore and Leonardo Quaresima (eds.): Il colore nel cinema muto. Bologna: Clueb, pp. 89–90. View Quote
Flueckiger, Barbara (2015 ): Color Analysis for the Digital Restoration of Das Cabinet des Dr. Caligari. In: Joshua Yumibe (ed.): Moving Image, 15/1, pp. 22-43 (peer-reviewed) Link to download
Street, Sarah; Yumibe, Joshua (2019): Chromatic Modernity. Color, Cinema, and Media of the 1920s. New York: Columbia University Press, on pp. 180–183. View Quote
Das idyllische Murg-Tal (GER 1910, Weltkinematograph):
Goergen, Jeanpaul (2010): Bunte Bilder aus dem Farbenbottich. Tonung und Virage in dokumentarischen Filmen der 1910er und 1920er Jahre. In: Filmblatt, pp. 3–52, on pp. 10–11. (in German) View Quote
Das Leben der Fischer in Concarneau (FRA 1911, Raleigh & Raleigh):
Goergen, Jeanpaul (2010): Bunte Bilder aus dem Farbenbottich. Tonung und Virage in dokumentarischen Filmen der 1910er und 1920er Jahre. In: Filmblatt, pp. 3–52, on pp. 11–12. (in German) View Quote
Der Harz, Nordhausen. Kulturfilm in 20 Abschnitten (GER 1922, Weltkinematograph):
Goergen, Jeanpaul (2010): Bunte Bilder aus dem Farbenbottich. Tonung und Virage in dokumentarischen Filmen der 1910er und 1920er Jahre. In: Filmblatt, pp. 3–52, on p. 39. (in German) View Quote
Die Ätna-Gürtelbahn (ITA 1910):
Goergen, Jeanpaul (2010): Bunte Bilder aus dem Farbenbottich. Tonung und Virage in dokumentarischen Filmen der 1910er und 1920er Jahre. In: Filmblatt, pp. 3–52, on p. 12. (in German) View Quote
Die Nibelungen (GER 1924, Fritz Lang):
Street, Sarah; Yumibe, Joshua (2019): Chromatic Modernity. Color, Cinema, and Media of the 1920s. New York: Columbia University Press, on pp. 173–180. View Quote
Die Sonne der Stadt (GER 1925, Elektrowerke AG):
Goergen, Jeanpaul (2010): Bunte Bilder aus dem Farbenbottich. Tonung und Virage in dokumentarischen Filmen der 1910er und 1920er Jahre. In: Filmblatt, pp. 3–52, on pp. 41–42. (in German) View Quote
Die Verräterin (GER 1911, Urban Gad):
De Kuyper, Eric (1992): La beauté du diable. In: Michel Ciment (ed.): Ciné mémoire. Colloque international d’information (7-9 octobre 1991). Paris: Femis, pp. 29–31, on p. 30. (in French) View Quote
Downhill (GBR 1927, Alfred Hitchcock) (sequence):
Street, Sarah (2012): Colour Films in Britain. The Negotiation of Innovation 1900-55. Basingstoke, Hampshire: Palgrave Macmillan, on pp. 19–20. View Quote
Il fuoco (ITA 1915, Giovanni Pastrone):
Pierotti, Federico (2012): La seduzione dello spettro. Storia e cultura del colore nel cinema. Genova: Le Mani-Microart, on pp. 72–73. (in Italian) View Quote
L’Atlantide/Queen of Atlantis (FRA/BEL 1921, Jacques Feyder):
De Kuyper, Eric (1992): La beauté du diable. In: Michel Ciment (ed.): Ciné mémoire. Colloque international d’information (7-9 octobre 1991). Paris: Femis, pp. 29–31, on pp. 30–31. (in French) View Quote
La poule aux œufs d’or (FRA 1905, Gaston Velle):
Pierotti, Federico (2012): La seduzione dello spettro. Storia e cultura del colore nel cinema. Genova: Le Mani-Microart, on pp. 43–47. (in Italian) View Quote
Ludwigslust im Film 1912-1914 (GER):
Goergen, Jeanpaul (2010): Bunte Bilder aus dem Farbenbottich. Tonung und Virage in dokumentarischen Filmen der 1910er und 1920er Jahre. In: Filmblatt, pp. 3–52, on pp. 14–15. (in German) View Quote
Napoléon (FRA 1927, Abel Gance):
Street, Sarah; Yumibe, Joshua (2019): Chromatic Modernity. Color, Cinema, and Media of the 1920s. New York: Columbia University Press, on pp. 1–2 View Quote and on pp. 217–219. View Quote
Traber, Bodo (1995): Dramaturgische Funktionen monochromer Farbgebung im Stummfilm. In: Karl-Dietmar Möller-Nass Möller-Nass, Hasko Schneider and Hans J. Wulff (eds.): 1. Film- und Fernsehwissenschaftliches Kolloquium. Münster: MAkS, pp. 30–36, on p. 35. (in German) View Quote
Neustadt in Sachsen: Im Zeichen des Schützenfestes (GER 1920, Heim-Licht):
Goergen, Jeanpaul (2010): Bunte Bilder aus dem Farbenbottich. Tonung und Virage in dokumentarischen Filmen der 1910er und 1920er Jahre. In: Filmblatt, pp. 3–52, on p. 40. (in German) View Quote
Rapsodia satanica (ITA 1917, Nino Oxilia):
Pierotti, Federico (2012): La seduzione dello spettro. Storia e cultura del colore nel cinema. Genova: Le Mani-Microart, on pp. 76–79. (in Italian) View Quote
The Affairs of Anatol (USA 1921, Cecil B. DeMille):
Fischer, Lucy (2017): Cinema by Design. Art Nouveau, Modernism and Film History. New York: Columbia University Press, on p. 76. View Quote
Street, Sarah; Yumibe, Joshua (2019): Chromatic Modernity. Color, Cinema, and Media of the 1920s. New York: Columbia University Press, on pp. 89–93. View Quote
The Great White Silence (GBR 1924, Herbert G. Ponting):
Watkins, Liz (2013): Herbert G. Ponting’s Materials and Texts. In: Simon Brown, Sarah Street and Liz Watkins (eds.): Color and the Moving Image. History, Theory, Aesthetics, Archive. New York, London: Routledge, pp. 230–242, on pp. 233–235. View Quote
The Lodger. A Story of the London Fog (GBR 1927, Alfred Hitchcock):
Street, Sarah; Yumibe, Joshua (2013): The Temporalities of Intermediality. Colour in Cinema and the Arts of the 1920s. In: Early Popular Visual Culture, 11,2, May, pp. 140–157, on pp. 152-154. View Quote
Street, Sarah; Yumibe, Joshua (2019): Chromatic Modernity. Color, Cinema, and Media of the 1920s. New York: Columbia University Press, on pp. 213-216. View Quote
The Phantom of the Opera (USA 1925, Rupert Julian):
Traber, Bodo (1995): Dramaturgische Funktionen monochromer Farbgebung im Stummfilm. In: Karl-Dietmar Möller-Nass Möller-Nass, Hasko Schneider and Hans J. Wulff (eds.): 1. Film- und Fernsehwissenschaftliches Kolloquium. Münster: MAkS, pp. 30–36, on pp. 31–33. (in German) View Quote
The Son of the Sheik (USA 1926, George Fitzmaurice):
Traber, Bodo (1995): Dramaturgische Funktionen monochromer Farbgebung im Stummfilm. In: Karl-Dietmar Möller-Nass Möller-Nass, Hasko Schneider and Hans J. Wulff (eds.): 1. Film- und Fernsehwissenschaftliches Kolloquium. Münster: MAkS, pp. 30–36, on pp. 32–35. (in German) View Quote
The Soul of the Cypress (USA 1920, Dudley Murphy):
Street, Sarah; Yumibe, Joshua (2019): Chromatic Modernity. Color, Cinema, and Media of the 1920s. New York: Columbia University Press, on p. 200. View Quote
The Ten Commandments (USA 1923, Cecil B. DeMille):
Birchard, Robert S. (1992): The Ten Commandments (1923). DeMille Completes Personal Exodus. In: American Cinematographer, 73,10, pp. 76–80, on pp. 76–77. View Quote
Layton, James; Pierce, David (2015): The Dawn of Technicolor. Rochester: George Eastman House, on pp. 99–102. View Quote
The Thief of Bagdad (USA 1924, Raoul Walsh):
Fischer, Lucy (2017): Cinema by Design. Art Nouveau, Modernism and Film History. New York: Columbia University Press, on p. 109. View Quote
Tigre reale (ITA 1916, Giovanni Pastrone):
Pierotti, Federico (2012): La seduzione dello spettro. Storia e cultura del colore nel cinema. Genova: Le Mani-Microart, on pp. 73–74. (in Italian) View Quote
Über Felsen und Eis (GER 1911, Weltkinematograph):
Goergen, Jeanpaul (2010): Bunte Bilder aus dem Farbenbottich. Tonung und Virage in dokumentarischen Filmen der 1910er und 1920er Jahre. In: Filmblatt, pp. 3–52, on p. 13. (in German) View Quote
Vorführung der ins Feld gehenden vom k.k. Kriegsministerium neu geschaffenen Feld-Sanitäts-Autokolonne (AUT 1915, Hans Otto Löwenstein, Sascha-Film):
Goergen, Jeanpaul (2010): Bunte Bilder aus dem Farbenbottich. Tonung und Virage in dokumentarischen Filmen der 1910er und 1920er Jahre. In: Filmblatt, pp. 3–52, on pp. 37–38. (in German) View Quote
Wie bleibe ich gesund? 1. Teil: Hygiene des häuslichen Lebens (GER 1922, Universum Film AG):
Goergen, Jeanpaul (2010): Bunte Bilder aus dem Farbenbottich. Tonung und Virage in dokumentarischen Filmen der 1910er und 1920er Jahre. In: Filmblatt, pp. 3–52, on p. 39. (in German) View Quote










































































